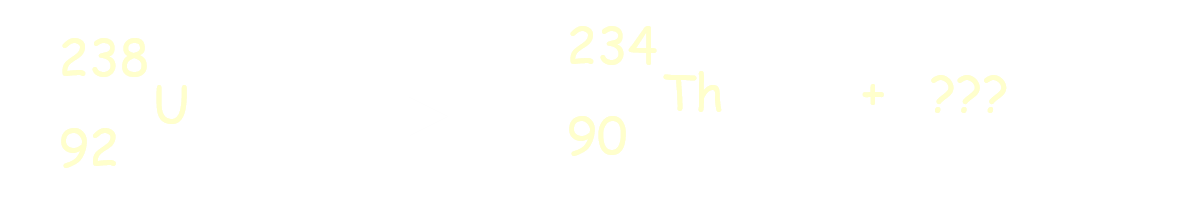Physical Science Nuclear Reactions
Presentation
•
Science
•
9th - 12th Grade
•
Medium
+13
Standards-aligned
Susan Schroer
Used 6+ times
FREE Resource
7 Slides • 10 Questions
1
2
Multiple Choice
3
Multiple Choice
4
5
6
7
Multiple Choice
8
9
10
11
Multiple Choice
12
Multiple Choice
13
Multiple Choice
14
Multiple Choice
15
Multiple Choice
16
Multiple Choice
17
Multiple Choice
Show answer
Auto Play
Slide 1 / 17
SLIDE
Similar Resources on Wayground

16 questions
Four Fundamental Forces
Lesson
•
9th - 12th Grade

14 questions
HR Diagram Test
Lesson
•
9th - 12th Grade

9 questions
Nuclear Energy from Kurzgesagt
Lesson
•
9th - 12th Grade

15 questions
Exponential vs. Linear: Growth and Decay
Lesson
•
9th - 12th Grade

14 questions
Solar Energies
Lesson
•
9th - 12th Grade

11 questions
protons, neutrons, and electrons
Lesson
•
KG - University

12 questions
The Solar System Intro
Lesson
•
10th - 12th Grade

17 questions
Convection Currents in the Mantle
Lesson
•
9th - 12th Grade
Popular Resources on Wayground

7 questions
History of Valentine's Day
Interactive video
•
4th Grade

15 questions
Fractions on a Number Line
Quiz
•
3rd Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

25 questions
Multiplication Facts
Quiz
•
5th Grade

22 questions
fractions
Quiz
•
3rd Grade

15 questions
Valentine's Day Trivia
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade
Discover more resources for Science

25 questions
Naming Ionic and Covalent Compounds
Quiz
•
9th Grade

20 questions
Mendelian Genetics Review
Quiz
•
9th Grade

10 questions
Exploring Weathering, Erosion, and Deposition Processes
Interactive video
•
6th - 10th Grade

10 questions
Exploring Mendelian Genetics and Punnett Squares
Interactive video
•
6th - 10th Grade

10 questions
Exploring Light and Waves Concepts
Interactive video
•
6th - 10th Grade

20 questions
The Cell Cycle and Mitosis
Quiz
•
9th Grade

35 questions
DNA Structure and Replication
Quiz
•
10th Grade

89 questions
Unit 1 (Ch 2 & 3) Test Review - Water/Ocean Currents
Quiz
•
9th - 12th Grade