

Rates of Reactions
Presentation
•
Science
•
9th - 12th Grade
•
Medium
Standards-aligned
Stacy Kowlsen
Used 33+ times
FREE Resource
12 Slides • 13 Questions
1

BELL RINGER
WATCH THE
VIDEO AND
RECORD
https://www.youtube.com/watch?v=OttRV
5ykP7A
2


ESSENTIAL
QUESTION
WHAT IS THE HEAT OF
REACTION (ΔH)?
3

STANDARD
SC.912.P.10.5
Use the kinetic-
molecular theory to
describe the behavior
and kinetic energy of a
molecule and compound
during changes in
temperature.
4

OBJECTIVES
Understand the 5
ways that speed up
a reaction rate and
why
Recognize what 2
things must happen
for a reaction to
happen
5

ENERGY AND
CHEMICAL
REACTIONS
Chemical Energy – Energy stored in
the chemical bonds of a substance.
Chemical reactions always involve
energy changes.
Making bonds and breaking bonds
involve energy changes
6

When you add heat energy, molecules
move faster; temperature increases.
• Temperature: a measure of heat,
increase heat, increase temperature
Chemical Potential Energy: stored
energy in chemical bonds
• Depends on the type of atoms and
arrangement
7


FACTORS THAT
INCREASE REACTION
RATES
Increasing surface area of a
solid reactant
Increasing temperature
Increasing concentration or
pressure of a reactant
Nature of the reactants
Adding a catalyst.
8



INCREASE SURFACE AREA
Increase surface area of the reactant by crushing solid reactant into smaller pieces.
Ex: Adding sugar to your Hot Tea
9


INCREASING TEMPERATURE
Increasing temperature increase kinetic energy which: (Learned in Gas Laws)
Breaks forces of attraction
Increases the number and intensity of collisions between reactants
10

INCREASE CONCENTRATION
OR PRESSURE
An increase in concentration means an increase in the amount of
reactant which will increase in the number of collisions between
the reactants which increases the rate of the reaction.
• An increase in
pressure will also
do the same thing
11

NATURE OF THE REACTANT
Individual properties of substances also affect reaction rates. Some of the properties in this
category are:
State of matter: Gases react faster than liquids and solids while aqueous tends to react fastest.
Molecular size/Number of bonds: Smaller molecules with less bonds react faster than bigger
molecules with more bonds.
Bond type: Ionic tend to react faster than covalent
Bond strength: Single bonds are easier and faster to break than double or triple bonds.
12


ADD A CATALYST
Activation Energy-the energy required for a
reaction to start. ALL CHEMICAL REACTIONS
REQUIRE ACTIVATION ENERGY!!!
Catalyst- is a substance that reduces the activation
energy of a reaction and causes the reaction to occur
“faster”. CATALYSTS ARE NOT PART OF THE
REACTION! They are unchanged during the reaction.
13
Multiple Choice
false
true
14
Multiple Choice
it increases both the frequency and energy of particle collisions
it only increases the frequency of particle collisions
it only increases the energy of particle collisions
it reduces the activation energy of the reaction
15
Multiple Choice
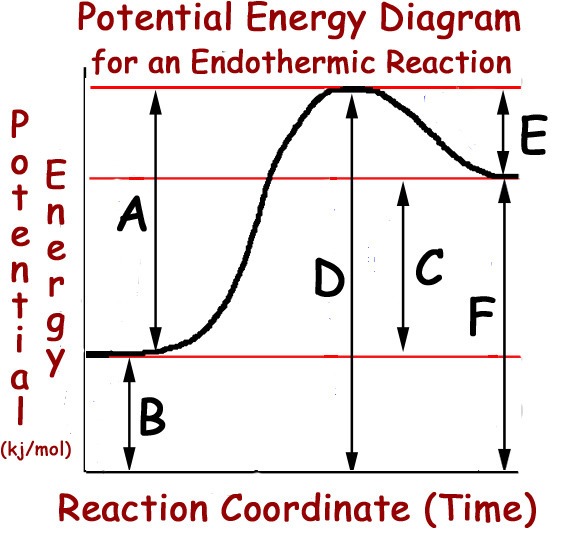
A
B
C
D
16
Multiple Choice
Why does a higher concentration increase the rate of reaction?
it increases the amount of reactants
it lowers the activation energy
it increases the energy of particle collisions
it increases the frequency of particle collisions
17
Multiple Choice
As the frequency of ______________ increases, the rate of reaction increases.
Time
Reactions
Collisions
Reactants
18
Multiple Choice
Adding a catalyst ___________ the activation energy.
Raises
Lowers
Doesn't affect
Inreases
19
Multiple Choice
The minimum amount of energy needed for colliding particles to react is called
Chemical Energy
Kinetic Energy
Activation Energy
Potential Energy
20
Multiple Choice

Powdered sugar has a greater ______ _____ than a solid cube of
sugar.
Surface Area
Catalyst
Temperature
21
Multiple Choice
sufficient rate and sufficient energy
sufficient surface area and correct orientation
sufficient catalyst and sufficient energy
sufficent energy and correct orientation
22
Multiple Choice

Which group of particles shows that increasing surface area increases collisions among particles?
Left side
Right side
Cannot be determined by either picture
23
Multiple Choice
larger
smaller
24
Multiple Choice
concentration
surface area
temperature
reactants
25
Multiple Choice
increasing temperature
increasing concentration
increasing surface area
all of these

BELL RINGER
WATCH THE
VIDEO AND
RECORD
https://www.youtube.com/watch?v=OttRV
5ykP7A
Show answer
Auto Play
Slide 1 / 25
SLIDE
Similar Resources on Wayground

19 questions
Unit 12- Work and Machines
Lesson
•
9th - 12th Grade

19 questions
Char of Life Review and Intro to Viruses and Bacteria
Lesson
•
9th - 12th Grade

18 questions
Impulse and Momentum
Lesson
•
9th - 12th Grade

21 questions
Chromosomal Mutations
Lesson
•
9th - 12th Grade

21 questions
1.9 Carbon
Lesson
•
9th - 12th Grade

20 questions
Introduction to Wave pt 3: EM vs Mechanical Waves
Lesson
•
9th - 12th Grade

19 questions
Biomes Intro
Lesson
•
9th - 12th Grade

19 questions
AP CSP Unit 6 Review 1
Lesson
•
9th - 12th Grade
Popular Resources on Wayground

10 questions
5.P.1.3 Distance/Time Graphs
Quiz
•
5th Grade

10 questions
Fire Drill
Quiz
•
2nd - 5th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

15 questions
Hargrett House Quiz: Community & Service
Quiz
•
5th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

15 questions
Equivalent Fractions
Quiz
•
4th Grade
Discover more resources for Science

10 questions
Exploring the Layers of the Earth
Interactive video
•
6th - 10th Grade

25 questions
Naming Ionic and Covalent Compounds
Quiz
•
9th Grade

20 questions
Balancing Chemical Equations
Quiz
•
9th Grade

35 questions
DNA Structure and Replication
Quiz
•
10th Grade

20 questions
Non-Mendelian Genetics
Quiz
•
9th Grade

10 questions
Exploring the Dynamics of Ocean Currents
Interactive video
•
6th - 10th Grade

20 questions
The Cell Cycle and Mitosis
Quiz
•
9th Grade

15 questions
Genetics- Punnett Squares
Quiz
•
9th Grade