

Nature of Matter Review
Presentation
•
Science
•
8th - 9th Grade
•
Medium
+4
Standards-aligned
Dana Wallach
Used 25+ times
FREE Resource
4 Slides • 20 Questions
1
Multiple Choice

When is the Unit 1 Test?
Never
Wednesday, Sept. 14
Thursday, Sept. 14
Friday, Sept. 16
2
Matter is made up of particles, called atoms.
These particles are in constant, random motion.
The state of matter depends on the kinetic energy (temperature) of the particles.
Kinetic Molecular Theory

As you increase in temperature, the substance goes from a solid, to a liquid, to a gas, and then a plasma (if it is hot enough).
3
Multiple Choice

Which of the following best represents three states (solid, liquid and gas) of matter?
Picture A
Picture B
Picture C
Picture D
4
Multiple Choice
As matter changes state from liquid to solid, the atoms slow down and move ______ .
CLOSER together
FARTHER apart
stay the SAME
5
Multiple Choice
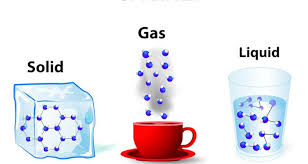
6
Multiple Choice
If I heat a liquid and its temperature increases, what change occurs to motion of the molecules?
The molecules move at the SAME rate.
The molecules move at a SLOWER rate.
The molecules move at a FASTER rate.
7
Multiple Choice

As matter changes state from gas to solid, the atoms have ____ kinetic energy.
more
less
the same
8
Physical properties of matter are properties that can be measured or observed without matter changing to an entirely different substance.
Physical properties

9
Chemical properties describe how a substance interacts with other substances to produce new substances.
Chemical properties

10
Multiple Choice

Physical properties are properties that can be observed
without changing the matter's identity.
that change the matter into new matter with new properties.
11
Multiple Choice
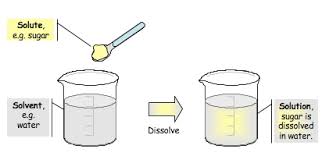
The ability of sugar to dissolve in water is called
malleability.
density.
state.
solubility.
12
Multiple Choice

Alkali earth metals react with hydrochloric acid.
chemical property
physical property
13
Multiple Choice

All of the following objects are attracted to magnets EXCEPT _________.
Iron nail
Plastic button
Steel scissors
Iron screw
14
Multiple Choice

A chemical property is a characteristic of a particular substance that can be observed in a chemical reaction.
True
False
15
Multiple Choice

Which of these objects is an insulator?
Copper Wire
Oven Mitt
Paper Clip
Soda Can
16
Multiple Choice
Which group of things will maintain their physical properties when mixed together?
copper wire, rubber ball and aluminum washer
Lemon juice, water and sugar
17
Multiple Choice

Identify which item below best completes the table.
Glass Window
Gold Ring
Paper Bag
Styrofoam Cup
18

Density is an important physical property of matter as it reflects how closely packed the particles of matter are. When particles are packed together more tightly, matter has greater density.
Density
19
Multiple Choice
The amount of matter in a given space of volume is
density.
state.
volume.
mass.
20
Multiple Choice

A sample of glass has a mass of 25 g and a volume of 10 cm3. What is the density of the sample?
1 g/cm3
2.5 g/cm3
25 g/cm3
250 g/cm3
21
Multiple Choice

A glass marble and a piece of cork of the same shape and size are dropped in water. Which of the following explains correctly why the glass marble sinks while the cork piece floats on water?
The glass marble is denser than water. The cork is less dense than water.
The glass marble is less dense than water. The cork is denser than water.
The glass marble and the cork are both denser than water.
The glass marble and the cork have the same density as water.
22
Multiple Choice

An unknown liquid has a mass of 10 gram and a volume of 10 mL. What is the density of the substance?
1 g/mL
10 g/mL
20 g/mL
100 g/mL
23
Multiple Choice

What is the least dense liquid in this container?
honey
vegetable oil
lamp oil
water
24
Match
Vocabulary Review - MATCH the following
mass
density
volume
matter
kinetic
amount of matter
mass per unit volume
amount of space an object occupies
anything that has volume and mass
relating to or resulting from motion
amount of matter
mass per unit volume
amount of space an object occupies
anything that has volume and mass
relating to or resulting from motion

When is the Unit 1 Test?
Never
Wednesday, Sept. 14
Thursday, Sept. 14
Friday, Sept. 16
Show answer
Auto Play
Slide 1 / 24
MULTIPLE CHOICE
Similar Resources on Wayground

20 questions
Weathering Erosion Deposition
Presentation
•
8th Grade

19 questions
Intro to Genetics
Presentation
•
9th Grade

19 questions
Eclipses
Presentation
•
8th Grade

19 questions
Skeleton & Bones Review
Presentation
•
9th Grade

19 questions
Periodic Table
Presentation
•
8th Grade

18 questions
Temperature
Presentation
•
8th Grade

18 questions
Bacteria and Viruses
Presentation
•
9th Grade

18 questions
Free-Body Diagrams
Presentation
•
7th - 9th Grade
Popular Resources on Wayground

10 questions
Factors 4th grade
Quiz
•
4th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

20 questions
Moon Phases and Eclipses
Quiz
•
6th - 8th Grade

45 questions
8th Grade Science - EOG Review
Quiz
•
8th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade

49 questions
NC Released EOG 8 Science (2024)
Quiz
•
8th Grade

48 questions
8th Grade NC Science EOG Review 2
Quiz
•
8th Grade

10 questions
Exploring Earth's Seasons and Their Causes
Interactive video
•
6th - 8th Grade