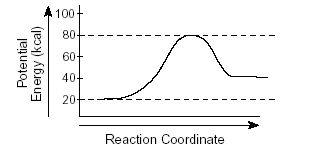
Final Exam Review-Part 1
Presentation
•
Chemistry
•
6th - 10th Grade
•
Easy
Annette Hall
Used 2+ times
FREE Resource
19 Slides • 29 Questions
1
2
3
Multiple Choice
4
5
Multiple Choice
6
7
Multiple Choice
8
Multiple Choice
9
10
Multiple Choice
11
Multiple Choice
12
Multiple Choice
13
Multiple Choice
14
Multiple Choice
15
16
Multiple Choice
17
Multiple Choice
18
19
20
Multiple Choice
21
22
Multiple Choice
23
Multiple Choice
24
Multiple Choice
25
Multiple Choice
26
Multiple Choice
27
Multiple Choice
28
Multiple Choice
29
30
Multiple Choice
31
32
Multiple Choice
33
34
Multiple Choice
35
36
Multiple Choice
37
Multiple Choice
38
Multiple Choice
39
Multiple Choice
40
41
42
43
44
Multiple Choice
45
46
Multiple Choice
47
Multiple Choice
48
Show answer
Auto Play
Slide 1 / 48
SLIDE
Similar Resources on Wayground

42 questions
Mga Katulong sa Komunidad Lesson
Lesson
•
KG

44 questions
G1 Sc CA 1 Revision
Lesson
•
KG

43 questions
Greenhouse Effect Notes
Lesson
•
6th - 10th Grade

44 questions
MonJ3++ L10 U6 Reading
Lesson
•
6th - 10th Grade

43 questions
Cell Cycle and Mitosis
Lesson
•
7th - 10th Grade

42 questions
RNA & Protein Synthesis
Lesson
•
7th - 9th Grade

44 questions
Primary Academic Refresher
Lesson
•
KG

38 questions
Rhetorical Appeals and Strategies
Lesson
•
9th Grade
Popular Resources on Wayground

15 questions
Fractions on a Number Line
Quiz
•
3rd Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

25 questions
Multiplication Facts
Quiz
•
5th Grade

29 questions
Alg. 1 Section 5.1 Coordinate Plane
Quiz
•
9th Grade

22 questions
fractions
Quiz
•
3rd Grade

11 questions
FOREST Effective communication
Lesson
•
KG

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade
Discover more resources for Chemistry

22 questions
Unit 9 Gas Law Quiz
Quiz
•
10th Grade

10 questions
Exploring Types of Chemical Reactions
Interactive video
•
6th - 10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Acids and Bases
Quiz
•
10th Grade

30 questions
Energy Review
Quiz
•
9th Grade

7 questions
GCSE Chemistry - Balancing Chemical Equations #4
Interactive video
•
9th - 10th Grade

20 questions
Chemistry: Classification of Matter
Quiz
•
10th Grade

40 questions
Unit 3 (Part 1) Chemical Equations & Reactions Review Game
Quiz
•
8th - 12th Grade