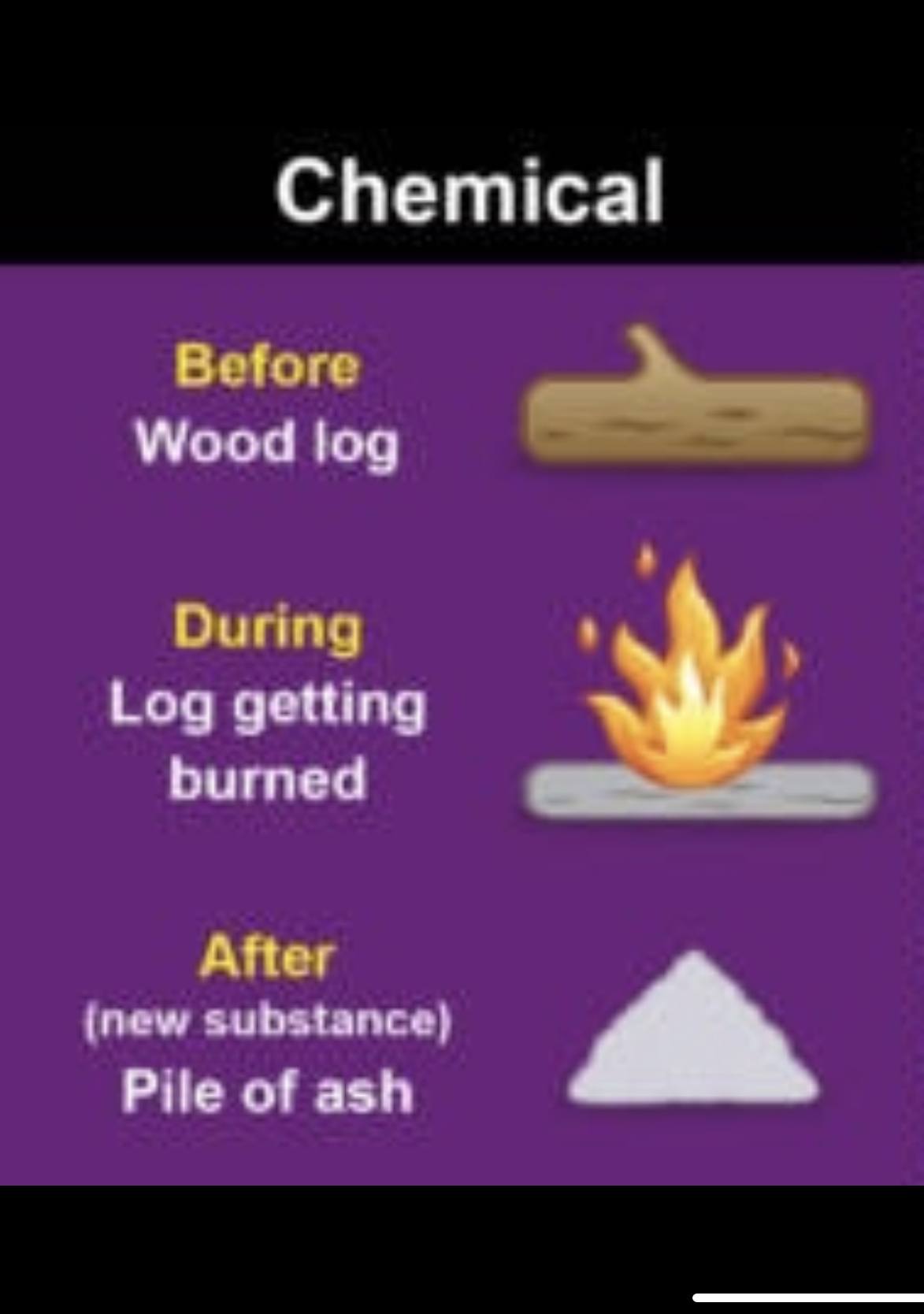
Physical and Chemical Changes
Presentation
•
Science
•
8th Grade
•
Medium
+1
Standards-aligned
Joseph F Mcguinness
Used 919+ times
FREE Resource
8 Slides • 5 Questions
1
2
Multiple Choice
3
4
5
6
Multiple Choice
7
Multiple Choice
8
9
10
11
Multiple Choice
12
13
Multiple Choice
Show answer
Auto Play
Slide 1 / 13
SLIDE
Similar Resources on Wayground

10 questions
Lesson 6: Acceleration
Lesson
•
8th Grade

13 questions
Speed, Velocity, Acceleration
Lesson
•
8th Grade

13 questions
Phase Change Diagrams
Lesson
•
8th Grade

11 questions
Dichotomous Key
Lesson
•
7th Grade

8 questions
Ellipsis and Dashes
Lesson
•
8th Grade

10 questions
Speed, Velocity, Acceleration-Practice
Lesson
•
7th Grade

9 questions
Energy types and transfers
Lesson
•
8th Grade

10 questions
Memory
Lesson
•
8th Grade
Popular Resources on Wayground

15 questions
Fractions on a Number Line
Quiz
•
3rd Grade

10 questions
Probability Practice
Quiz
•
4th Grade

15 questions
Probability on Number LIne
Quiz
•
4th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

25 questions
Multiplication Facts
Quiz
•
5th Grade

22 questions
fractions
Quiz
•
3rd Grade

6 questions
Appropriate Chromebook Usage
Lesson
•
7th Grade

10 questions
Greek Bases tele and phon
Quiz
•
6th - 8th Grade
Discover more resources for Science

10 questions
Exploring the Rock Cycle
Interactive video
•
6th - 8th Grade

8 questions
Newton's Second Law
Lesson
•
6th - 8th Grade

8 questions
Amoeba Sister Asexual vs Sexual Reproduction
Interactive video
•
8th Grade

10 questions
Exploring the Rock Cycle: Types and Formation
Interactive video
•
6th - 8th Grade

20 questions
Plate Tectonics
Quiz
•
8th Grade

25 questions
Evolution and Natural Selection Review
Quiz
•
6th - 8th Grade

10 questions
Exploring Weathering, Erosion, and Deposition Processes
Interactive video
•
6th - 10th Grade

20 questions
Rock Cycle
Quiz
•
8th Grade